A while ago I drove from Denver to Fort Worth, and I was impressed. See, there’s a lot of not much in eastern Colorado. It’s pretty much the same in western Oklahoma except there’s less not much because there’s less of Oklahoma – but Texas has way more not much than anybody.
That gives Texas not much to brag about, but they do the best they can, bless their hearts.
What got me started on this rant was a a pair of astronomical factoids Katherine Kornei wrote in the Nov 2014 Discover magazine.
“If galaxies were shrunk to the size of apples, neighboring galaxies would be only a few meters apart….”
“If the stars within galaxies were shrunk to the size of oranges, they would be separated by 4,800 kilometers (3,000 miles).”
 So there’s a lot of not much between galaxies, but a whole lot more not much, relatively speaking, within them. I just measured an apple and an orange in my kitchen. They’re both about the same size, 3 inches in diameter, so I have no idea why she chose different fruits – perhaps she wanted to avoid comparing apples and oranges.
So there’s a lot of not much between galaxies, but a whole lot more not much, relatively speaking, within them. I just measured an apple and an orange in my kitchen. They’re both about the same size, 3 inches in diameter, so I have no idea why she chose different fruits – perhaps she wanted to avoid comparing apples and oranges.
Anyway, if you felt like doing the galaxy visualization you could put two apple galaxies on the floor about 12 feet apart and then line up about 50 apples between them. A fair amount of space for more galaxies.
To see inside a galaxy you could put one orange star in Miami FL, and its on-the-average nearest orange neighbor in Seattle WA. Then you could set out a long skinny row of just about 63 million oranges in between. Oh, and on this scale the nearest galaxy would be about 2 billion miles (or 43 quadrillion oranges) away. Way more not much inside a galaxy than between two neighboring ones.
So if we squeeze all those apples and oranges together we’d get rid of all the empty space, right?
Not by a long shot. Nearly all those stars are balls of very hot gas, which means they’re made up of atoms crossing empty space inside the star to collide with other atoms. Relative to the size of the atoms, how much empty space is there inside the star?
 For example, every chemistry student learns that 6×1023 molecules of any gas take up a volume of 22.4 liters at normal Earth temperature and pressure. For a single-atom gas like helium that works out to about 22 atom-widths between atoms.
For example, every chemistry student learns that 6×1023 molecules of any gas take up a volume of 22.4 liters at normal Earth temperature and pressure. For a single-atom gas like helium that works out to about 22 atom-widths between atoms.
Now think about emptiness inside the Sun. If it’s a typical star (which it is) and if all of its atoms are hydrogen (which they mostly are) and if the average density of the Sun (1408 kg/m3) applied all the way down to the center of the Sun (which it doesn’t), and if we believe NASA’s numbers for the Sun (hey, why not?), then the average density works out to about 0.7 atom-widths between neighbors.
So no empty space to squeeze out of the Sun, eh? Well, actually there is quite a lot, because those atoms are mostly empty space, too.
OK, I cheated up there about the Sun, because virtually all of the Sun’s atoms have been dissociated into separated electrons and nuclei. The nucleus is much smaller than than its atom – by a factor of 60,000 or so. Think of a grape seed in the middle of a football field.
To sum it upward, we’ve got a set of Russian matryoshka dolls, one inside the next. At the center is a collection of grape seeds, billions and billions of them, each in their own football field. The football fields are all balled into a stellar orange (or maybe an apple), but there are billions of those crammed into a galactic apple (or maybe an orange) that’s about ten feet away from the nearest other piece of fruit.
As Douglas Adams wrote in Hitchhiker’s Guide to The Galaxy,
“Space … is big. Really big. You just won’t believe how vastly, hugely, mindbogglingly big it is. I mean, you may think it’s a long way down the road to the chemist’s, but that’s just peanuts to space…”
The thing to realize is that the function of all that space is to keep everything from being in the same place. That’s important.
~~ Rich Olcott




 Newton definitely didn’t see that one coming. He has an excuse, though. No-one in in the 17th Century even realized that electricity is a thing, much less that the electrostatic force follows the same inverse-square law that gravity does. So there’s no way poor Isaac would have come up with quantum mechanics.
Newton definitely didn’t see that one coming. He has an excuse, though. No-one in in the 17th Century even realized that electricity is a thing, much less that the electrostatic force follows the same inverse-square law that gravity does. So there’s no way poor Isaac would have come up with quantum mechanics. If Newton loved anything (and that question has been discussed at length), he loved an argument. His battle with Leibniz is legendary. He even fought with Descartes, who was a decade dead when Newton entered Cambridge.
If Newton loved anything (and that question has been discussed at length), he loved an argument. His battle with Leibniz is legendary. He even fought with Descartes, who was a decade dead when Newton entered Cambridge.
 in his Les Miz role of Inspector Javert,
in his Les Miz role of Inspector Javert, 
 And then there’s
And then there’s 
 Keep going until the outermost hexagon has 32 dots along each edge. All the hexagons together will have exactly 2016 dots.
Keep going until the outermost hexagon has 32 dots along each edge. All the hexagons together will have exactly 2016 dots.
 Newton was essentially a geometer. These illustrations (from Book 1 of the Principia) will give you an idea of his style. He’d set himself a problem then solve it by constructing sometimes elaborate diagrams by which he could prove that certain components were equal or in strict proportion.
Newton was essentially a geometer. These illustrations (from Book 1 of the Principia) will give you an idea of his style. He’d set himself a problem then solve it by constructing sometimes elaborate diagrams by which he could prove that certain components were equal or in strict proportion. For instance, in the first diagram (Proposition II, Theorem II), we see an initial glimpse of his technique of successive approximation. He defines a sequence of triangles which as they proliferate get closer and closer to the curve he wants to characterize.
For instance, in the first diagram (Proposition II, Theorem II), we see an initial glimpse of his technique of successive approximation. He defines a sequence of triangles which as they proliferate get closer and closer to the curve he wants to characterize. The third diagram is particularly relevant to the point I’ll finally get to when I get around to it. In Prop XLIV, Theorem XIV he demonstrates something weird. Suppose two objects A and B are orbiting around attractive center C, but B is moving twice as fast as A. If C exerts an additional force on B that is inversely dependent on the cube of the B-C distance, then A‘s orbit will be a perfect circle (yawn) but B‘s will be an ellipse that rotates around C, even though no external force pushes it laterally.
The third diagram is particularly relevant to the point I’ll finally get to when I get around to it. In Prop XLIV, Theorem XIV he demonstrates something weird. Suppose two objects A and B are orbiting around attractive center C, but B is moving twice as fast as A. If C exerts an additional force on B that is inversely dependent on the cube of the B-C distance, then A‘s orbit will be a perfect circle (yawn) but B‘s will be an ellipse that rotates around C, even though no external force pushes it laterally.



 It all started with Newton’s mechanics, his study of how objects affect the motion of other objects. His vocabulary list included words like force, momentum, velocity, acceleration, mass, …, all concepts that seem familiar to us but which Newton either originated or fundamentally re-defined. As time went on, other thinkers added more terms like power, energy and action.
It all started with Newton’s mechanics, his study of how objects affect the motion of other objects. His vocabulary list included words like force, momentum, velocity, acceleration, mass, …, all concepts that seem familiar to us but which Newton either originated or fundamentally re-defined. As time went on, other thinkers added more terms like power, energy and action.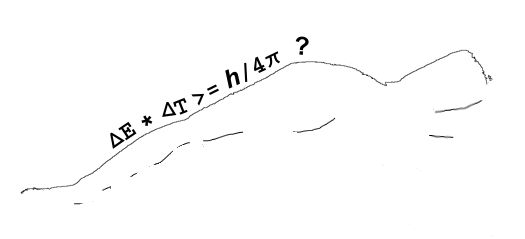 There is another way to get the same dimension expression but things aren’t not as nice there as they look at first glance. Action is given by the amount of energy expended in a given time interval, times the length of that interval. If you take the product of energy and time the dimensions work out as (ML2/T2)*T = ML2/T, just like Heisenberg’s Area.
There is another way to get the same dimension expression but things aren’t not as nice there as they look at first glance. Action is given by the amount of energy expended in a given time interval, times the length of that interval. If you take the product of energy and time the dimensions work out as (ML2/T2)*T = ML2/T, just like Heisenberg’s Area.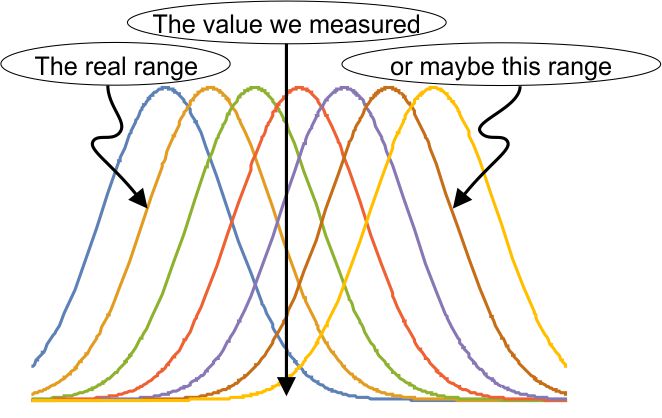
 For practice using Heisenberg’s Area, what can we say about the atom? (If you’re checking my math it’ll help to know that the Area, h/4π, can also be expressed as 0.5×10-34 kg m2/s; the mass of one hydrogen atom is 1.7×10-27 kg; and the speed of light is 3×108 m/s.) On average the atom’s position is at the cube’s center. Its position range is one meter wide. Whatever the atom’s average momentum might be, our measurements would be somewhere within a momentum range of (h/4π kg m2/s) / (1 m) = 0.5×10-34 kg m/s. A moving particle’s momentum is its mass times its velocity, so the velocity range is (0.5×10-34 kg m/s) / (1.7×10-27 kg) = 0.3×10-7 m/s.
For practice using Heisenberg’s Area, what can we say about the atom? (If you’re checking my math it’ll help to know that the Area, h/4π, can also be expressed as 0.5×10-34 kg m2/s; the mass of one hydrogen atom is 1.7×10-27 kg; and the speed of light is 3×108 m/s.) On average the atom’s position is at the cube’s center. Its position range is one meter wide. Whatever the atom’s average momentum might be, our measurements would be somewhere within a momentum range of (h/4π kg m2/s) / (1 m) = 0.5×10-34 kg m/s. A moving particle’s momentum is its mass times its velocity, so the velocity range is (0.5×10-34 kg m/s) / (1.7×10-27 kg) = 0.3×10-7 m/s. elaborate mathematical structure. If the measurement is a quantum mechanical result, part of that structure is our familiar bell-shaped curve. It’s an explicit recognition that way down in the world of the very small, we can’t know what’s really going on. Most calculations have to be statistical, predicting an average and an expected range about that average. That prediction may or may not pan out, depending on what the experimentalists find.
elaborate mathematical structure. If the measurement is a quantum mechanical result, part of that structure is our familiar bell-shaped curve. It’s an explicit recognition that way down in the world of the very small, we can’t know what’s really going on. Most calculations have to be statistical, predicting an average and an expected range about that average. That prediction may or may not pan out, depending on what the experimentalists find. So there could be a collection of bell-curves gathered about the experimental result. Remember those
So there could be a collection of bell-curves gathered about the experimental result. Remember those 






