Vinnie blows into Cal’s place dripping wet. “Man, it’s coming down out there! This monsoon’s trying to drown us.”
“No monsoon, Vinnie, this rain’s from an atmospheric river.”
“Monsoon, river, what’s the difference, I’m soaked, Cathleen!”
“Cal, Vinnie needs a warm‑up mug, quick!”
“Comin’ right up. Here you go, Vinnie. Cathleen, I’ve always used ‘monsoon’ for rain like this. What’s wrong with that?”
“Because a proper monsoon is seasonal; the year’s too early for monsoon season around here.”
“Didn’t realize we had a monsoon season. I just figure if it’s not snowing and it’s not steaming we got rain.”
“Not funny, Vinnie. Our local weather deserves better from you. We have lots of sunny days even during our July‑September monsoon season.”
“Why pick on those particular months?”

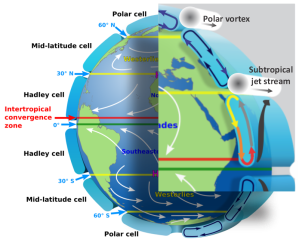
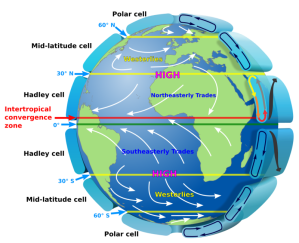
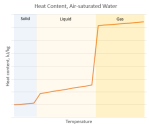
“Temperature and wind patterns. Sy would push the ‘because’ all the way down to heat capacity difference. Per unit area, a one‑centimeter layer of rock has about ¾ the heat capacity of the same thickness of still water. Convective water circulation magnifies the difference by ten.”
“So?”
“So a given amount of energy will raise the temperature of a rock expanse almost 13 times as many degrees as it would the same ocean area. On the other hand, night‑time energy radiation cools rock by almost 13 degrees where the ocean would cool by one. Compared to the ocean, land surface gets hotter in the daytime, cools off more at night.”
“So?”

“So Summer sunlight heats land area more than its neighboring ocean. Air over warm land expands and rises, making room for water‑laden air from the relatively cool ocean cover. That air warms and rises, too, but as it rises it cools again. The water vapor condenses and rain falls. Come Autumn, land temperature drops more than ocean surface temperature does. That reverses the air’s general circulation and shuts off the rain. End of monsoon season.”
“It works that way all over the world?”
“Most everywhere except Antarctica. Apparently it stays so cold there all year around that the back‑and‑forth rocking never gets a chance to develop.”
“Doesn’t seem to have anything to do with the Coriolis Effect.”
“Most of the movement is roughly perpendicular to the local coastlines, but Coriolis gets into play farther away from the coast. The North American monsoon, for instance, comes up from the Gulf of California and trends northeast across the mountain states into Kansas and Oklahoma.”
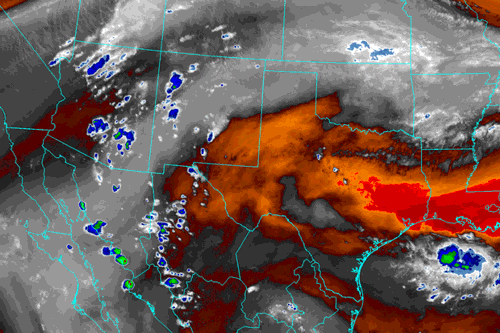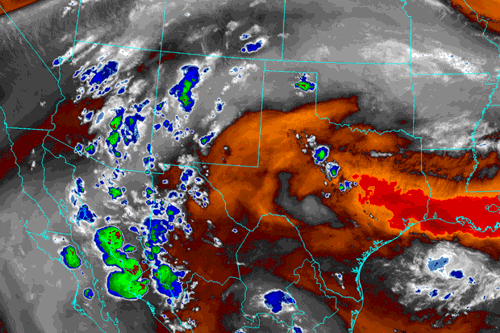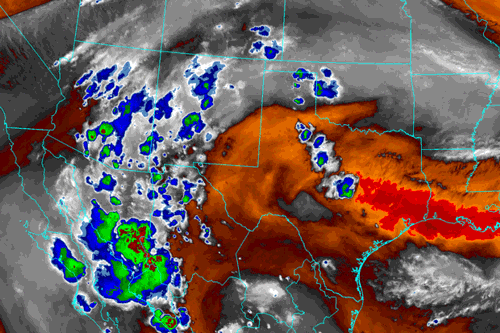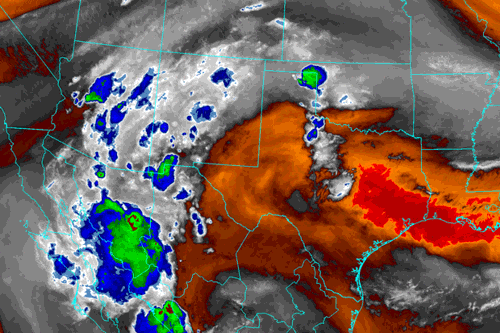
Image from Albuquerque, NM National Weather Service
“That strong west‑to‑east trend looks like some weather maps I’ve used flown by. Is that an atmospheric river?”
“Similar, Vinnie, but no. Atmospheric rivers are a completely separate phenomenon from monsoons — different physical settings, different behavior. They’re both fueled by warm oceanic water vapor but that’s about it. Monsoons are seasonal, last for months, and hover over particular regions; atmospheric rivers are perennial, generally live for only a few days, and wander around. We’ve known about monsoons for millennia, but it wasn’t until 1992 that scientists had satellite data to support the rivers notion.”
“How can they be perennial and only last for days?”
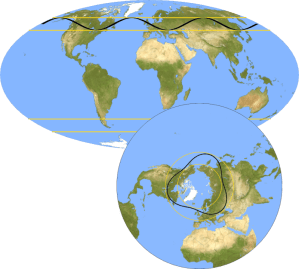
“I meant that individual rivers come and go but the northern and southern hemispheres generally have about four each throughout the year. Rivers thousands of kilometers long, hundreds of kilometers wide, carrying as much water as the Amazon. Together they’re responsible for transporting 80‑90% of the tropical moisture that crosses the mid‑latitudes each year.”
“That’s a lot of water hanging over our heads. How could people have just missed them all those years?”
Vinnie has a calculating look. “A million square kilometers. How tall are these things?”
“Can’t give you a firm answer. Probably a kilometer or two but that’s a guess. The satellite sensors register total water mass from orbit down to ground. The numbers peak around 60 millimeters of H₂O per square centimeter. Spread along a one‑kilometer height that’s 60 g/m³ maximum; figure a 30‑gram average.”
“It’s just moist air, then, not liquid water. Cal, your breath’s more humid than that. A million cubic kilometers of 30‑gram 1‑meter cubes packs a lot of grams. I’m sure I’ve flown right through those things without noticing them.”
~ Rich Olcott