Amazing what you can do with mozzarella drips and crumbled pizza edges. Vinnie’s rolling his crumbs into decent-sized marbles. (Pizza-maker Eddie’s giving him a look.) He adds a fourth ball to his triangle to make a square. “So anyway, what you’re telling us is that Bohr’s 8-electron shell isn’t that far off.”
“Oh, it is far off. Bohr put his electrons in a plane like your square there. Try putting that fourth ball on top of the others to make a triangular pyramid. See that? Counting the bottom it’s a four-sided figure called a tetrahedron. It’s the fundamental structural building block for most of the Universe’s molecules.” “Hey, that’s the alpha-particle shape that the protons and neutrons get themselves into.”
“Hey, that’s the alpha-particle shape that the protons and neutrons get themselves into.”
“Good point, Vinnie. Mind you, though, an alpha particle doesn’t have a central attractor, and it’s a quarter-million times smaller than an atom’s electron cloud. Got that pyramid shape in mind?”
“Sure.”
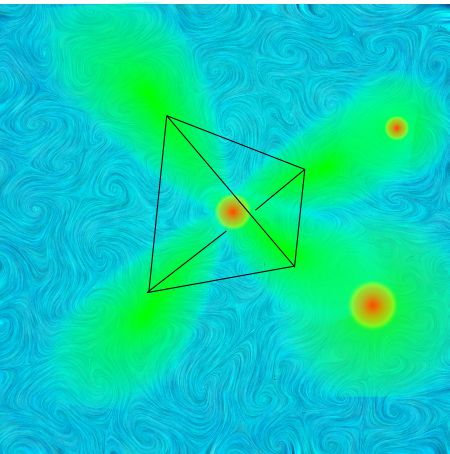
“OK, put those balls back in your square. … Put a finger on the north ball and another on the south one. Now roll them both up into contact on top of the line between the east and west ones.”
“Hey, it’s that tetra-thing again.”
“Right, Eddie. Any time you have four objects each the same distance from all the others, you’ve got a tetrahedron. If the ‘objects’ are clouds of electron charge all attracted to the nucleus and all repelled by the other clouds, that’s the shape they’ll take. It’s no accident that an equal mix of an atom’s spherical and three dumbbell orbitals in a shell makes four equivalent orbitals pointing to the corners of a tetrahedron.”
“Cute, but what’s it get us?”
“It gets us to the chemists’ trick for thinking about molecular structures without doing all the quantum mechanics. The key is that 8-electron shell. Forget electrons racing in a ring or electron pairs in a square. When you see a chemical diagram with four lines coming out of a central atom, think of them in a tetrahedron. Here’s an example. Guess what’s the commonest atom in the Universe.”
“Helium.”
“Hydrogen.”
“Eddie’s win with hydrogen — 923,000 atoms out of a million. Carbon’s the fourth most common, 480 atoms per million. Think of a carbon atom, floating around in space with four of its six units of electronic charge in its 2-shell. And it’s surrounded by hydrogen atoms with electrons just begging to pair up with something. No surprise, there’s suddenly a lot of electron pairing and you’ve got a molecule of methane, CH4. What’s its shape? Any hydrogen-hydrogen chains in there?”
“With this build-up, I gotta guess they’re all on the carbon and that they’re splayed out tetrahedron-like, hydrogen centers trying to get away from the other ones and shared charge clouds trying to get away from each other, too.”
“Couldn’t put it better myself, Vinnie.”
“Hey, water’s H2O, right? You can’t make a tetrahedron from only three atoms.”
“True, Eddie, but an oxygen atom comes with two more electrons than carbon has. We’ve still got a tetrahedron, but only two of its corners carry a hydrogen. The other two orbitals stick out their own directions, each loaded with negative charge. The chemists call that unshared kind of orbital a lone pair. They often show it as a double-dot on the structure diagram. That’s basically just a bookkeeping device to keep track of electron counts. All the charge is really spread around throughout all the molecular orbitals just like with atomic orbitals, only it’s not spread evenly.”
“Why do they bother to keep track like that?”
“Lone pairs affect the molecule’s structure. If it weren’t for them, the water molecule would be a straight line. In fact, a lone pair orbital crowds the space a bit more than a bonding pair — the H–O–H angle is about 5º smaller than a perfect tetrahedron.”
“Makes sense when you think about it, like you can wave a stick all over the place unless someone grabs the other end.”
“Mm-hm. The big reason chemists care, though, is that lone pairs can be active centers during a chemical reaction. All that negative charge just waiting for something positive-ish to come along.”
“Like a really good tip,” grumbles Eddie.
~~ Rich Olcott










 . Z is the nuclear charge, which they’d just figured out how to measure, and R is a constant. Funny how it just happens to be Rydberg’s initial.”
. Z is the nuclear charge, which they’d just figured out how to measure, and R is a constant. Funny how it just happens to be Rydberg’s initial.”
 “But GR’s not the only player. Special Relativity’s in there, too.”
“But GR’s not the only player. Special Relativity’s in there, too.”




