 Newton definitely didn’t see that one coming. He has an excuse, though. No-one in in the 17th Century even realized that electricity is a thing, much less that the electrostatic force follows the same inverse-square law that gravity does. So there’s no way poor Isaac would have come up with quantum mechanics.
Newton definitely didn’t see that one coming. He has an excuse, though. No-one in in the 17th Century even realized that electricity is a thing, much less that the electrostatic force follows the same inverse-square law that gravity does. So there’s no way poor Isaac would have come up with quantum mechanics.
Lemme ‘splain. Suppose you have a mathematical model that’s good at predicting some things, like exactly where Jupiter will be next week. But if the model predicts an infinite value under some circumstances, that tells you it’s time to look for a new model for those particular circumstances.
For example, Newton’s Law of Gravity says that the force between two objects is proportional to 1/r2, where r is the distance between their centers of mass. The Law does a marvelous job with stars and satellites but does the infinity thing when r approaches zero. In prior posts I’ve described some physics models that supercede Newton’s gravity law at close distances.
Electrical forces are same song second verse with a coda. They follow the 1/r2 law, so they also have those infinity singularities. According to the force law, an electron (the ultimate “particle” of negative charge) that approaches another electron would feel a repulsion that rises to infinity. The coda is that as an electron approaches a positive atomic nucleus it would feel an attraction that rises to infinity. Nature abhors infinities, so something else, some new physics, must come into play.
I put that word “particle” in quotes because common as the electron-is-a-particle notion is, it leads us astray. We tend to think of the electron as this teeny little billiard-ballish thing, but it’s not like that at all. It’s also not a wave, although it sometimes acts like one. “Wavicle” is just a weasel-word. It’s far better to think of the electron as just a little traveling parcel of energy. Photons, too, and all those other denizens of the sub-atomic zoo.
An electron can’t crumble or leak mass or deform to merge the way that sizable objects can. What it does is smear. Quantum mechanics is all about the smear. Much more about that in later posts.
 If Newton loved anything (and that question has been discussed at length), he loved an argument. His battle with Leibniz is legendary. He even fought with Descartes, who was a decade dead when Newton entered Cambridge.
If Newton loved anything (and that question has been discussed at length), he loved an argument. His battle with Leibniz is legendary. He even fought with Descartes, who was a decade dead when Newton entered Cambridge.
Descartes had grabbed “Nature abhors a vacuum” from Aristotle and never let it go. He insisted that the Universe must be filled with some sort of water-like fluid. He know the planets went round the Sun despite the fluid getting in the way, so he reasoned they moved as they did because of the fluid.
Surely you once played with toy boats in the bathtub. You may have noticed that when you pulled your arm quickly through the water little whirlpools followed your arm. If a whirlpool encountered a very small boat, the boat might get caught in it and move in the same direction. Descartes held that the Solar System worked like that, with the Sun as your arm and the planets caught in Sun-stirred vortices within that watery fluid.
Newton knew that couldn’t be right. The planets don’t run behind the Sun, they share the same plane. Furthermore, comets orbit in from all directions. Crucially, Descartes’ theory conflicted with his own and that settled the matter for Newton. Much of Principia‘s “Book II” is about motions of and through fluid media. He laid out there what a trajectory would look like under a variety of conditions. As you’d expect, none of the paths do what planets, moons and comets do.
From Newton’s point of view, the only use for Book II was to demolish Descartes. For us in later generations, though, he’d invented the science of hydrodynamics.
Which was a good thing so long as you don’t go too far upstream towards the center of the whirlpool. As you might expect (or I wouldn’t even be writing this section), Book II is littered with 1/rn formulas that go BLOOIE when the distances get short. What happens near the center? That’s where the new physics of turbulence kicks in.
~~ Rich Olcott




 It all started with Newton’s mechanics, his study of how objects affect the motion of other objects. His vocabulary list included words like force, momentum, velocity, acceleration, mass, …, all concepts that seem familiar to us but which Newton either originated or fundamentally re-defined. As time went on, other thinkers added more terms like power, energy and action.
It all started with Newton’s mechanics, his study of how objects affect the motion of other objects. His vocabulary list included words like force, momentum, velocity, acceleration, mass, …, all concepts that seem familiar to us but which Newton either originated or fundamentally re-defined. As time went on, other thinkers added more terms like power, energy and action.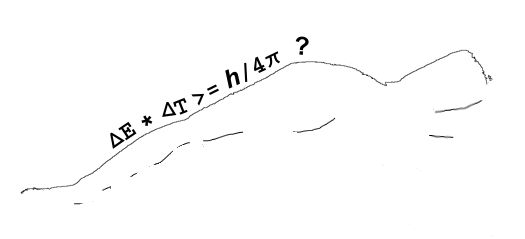 There is another way to get the same dimension expression but things aren’t not as nice there as they look at first glance. Action is given by the amount of energy expended in a given time interval, times the length of that interval. If you take the product of energy and time the dimensions work out as (ML2/T2)*T = ML2/T, just like Heisenberg’s Area.
There is another way to get the same dimension expression but things aren’t not as nice there as they look at first glance. Action is given by the amount of energy expended in a given time interval, times the length of that interval. If you take the product of energy and time the dimensions work out as (ML2/T2)*T = ML2/T, just like Heisenberg’s Area.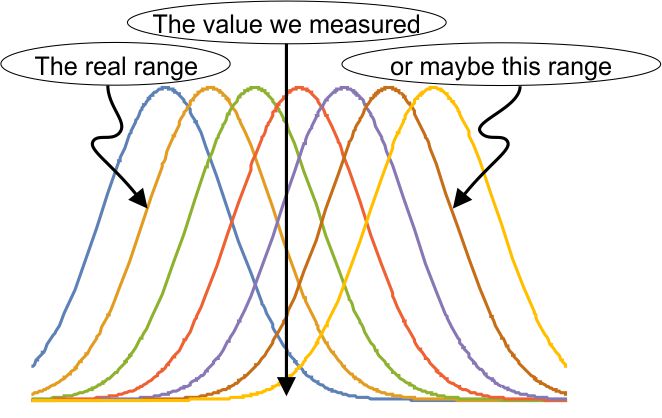
 For practice using Heisenberg’s Area, what can we say about the atom? (If you’re checking my math it’ll help to know that the Area, h/4π, can also be expressed as 0.5×10-34 kg m2/s; the mass of one hydrogen atom is 1.7×10-27 kg; and the speed of light is 3×108 m/s.) On average the atom’s position is at the cube’s center. Its position range is one meter wide. Whatever the atom’s average momentum might be, our measurements would be somewhere within a momentum range of (h/4π kg m2/s) / (1 m) = 0.5×10-34 kg m/s. A moving particle’s momentum is its mass times its velocity, so the velocity range is (0.5×10-34 kg m/s) / (1.7×10-27 kg) = 0.3×10-7 m/s.
For practice using Heisenberg’s Area, what can we say about the atom? (If you’re checking my math it’ll help to know that the Area, h/4π, can also be expressed as 0.5×10-34 kg m2/s; the mass of one hydrogen atom is 1.7×10-27 kg; and the speed of light is 3×108 m/s.) On average the atom’s position is at the cube’s center. Its position range is one meter wide. Whatever the atom’s average momentum might be, our measurements would be somewhere within a momentum range of (h/4π kg m2/s) / (1 m) = 0.5×10-34 kg m/s. A moving particle’s momentum is its mass times its velocity, so the velocity range is (0.5×10-34 kg m/s) / (1.7×10-27 kg) = 0.3×10-7 m/s. elaborate mathematical structure. If the measurement is a quantum mechanical result, part of that structure is our familiar bell-shaped curve. It’s an explicit recognition that way down in the world of the very small, we can’t know what’s really going on. Most calculations have to be statistical, predicting an average and an expected range about that average. That prediction may or may not pan out, depending on what the experimentalists find.
elaborate mathematical structure. If the measurement is a quantum mechanical result, part of that structure is our familiar bell-shaped curve. It’s an explicit recognition that way down in the world of the very small, we can’t know what’s really going on. Most calculations have to be statistical, predicting an average and an expected range about that average. That prediction may or may not pan out, depending on what the experimentalists find. So there could be a collection of bell-curves gathered about the experimental result. Remember those
So there could be a collection of bell-curves gathered about the experimental result. Remember those 








