<from the casebook of Sy Moire, Consulting Physicist> Monday. Weather sunny, warm for this time of year. Dry, bad effects on nose and throat at wake‑up time. Bought a room humidifier Friday at Big Box — up‑scale, 4‑liter reservoir, ultrasonic but silent, WiFi‑enabled etc. Long way from the jug with boiler tube my folks used to use. WiFi’s a sneaky way to avoid building a remote control — just use the customer’s smart phone. Guess that keeps the price down.
Good news is, phone app does graphs of relative humidity against time. Had it in measure-only mode in office Monday night, baseline wobbled in the 32-36% range. Bad news is, when the device is running on Automatic it works toward a 45% target but had trouble getting near that high on its maiden effort on Saturday. Wondered, how much water would it have to send into the room to hit the target?
Start with the numbers. “45%” is forty‑five percent of what? Surely not some arbitrary maximum. Weather guy talks about relative humidity so, relative to what? Searched a little in the internet. All the sources say RH is a ratio of ratios, something over maximum something. “Something” is water vapor mass per unit volume or else water vapor mass per total gas mass in the same volume. Clouds, fog, raindrops and snowflakes don’t count. Then things get fuzzy. Some sources say the maximum is “saturation” which is just a tautology. The most precise definition says “the partial pressure of water in air over a large flat surface of pure water under laboratory conditions” <shudder>.
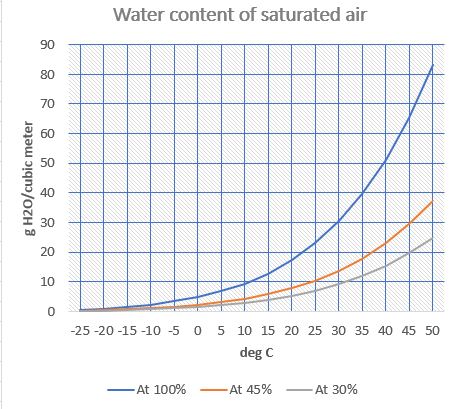
Doing engineer stuff here so keep it simple. Found a chart of water vapor content at 100% humidity at different temperatures. Will go with that. No surprise, the warmer the air, the more water mass it can hold before fogging up. I’m comfortable around 68°F which is 20°C. The chart says 100% saturated 20°C air holds 17.3 grams of water per cubic meter. 45% of that is 7.8 and 30% is 5.3. Need to know how many cubic meters in the office.
OK, measured the room as 10’×15′ with an 8′ ceiling. Ignore the space the furniture takes up. Total volume is 1200 cubic feet. Old Reliable says that’s 34 cubic meters. If the room’s at 100% humidity its air holds
(34 m3)×(17.3 g/m3)
= 588 grams of water.
At 1000 grams per liter that’s 0.588 liter or about a pint. Suppose humidifier starts when the room’s at 30% humidity. For a 15% bump to 45% the gadget has to vaporize
(34 m3)×(17.3 g/m3)×(0.45-0.30)
= 88 g = 88 milliliters.
Wow, that’s only about 6 tablespoons. Does that number even make sense? OK, air is about 80% N2, molecular weight 28. The other 20% is mostly O2, molecular weight 32. The average is near 29. Basic chem class stuff — at Earth‑typical room temp and pressure, a 22.4‑liter chunk of air has a mass near 29 grams, so office’s roomful of air would mass
(29g/mole)×(1 mole/22.4 liters)
×(1000 liters/m3)×(34 m3/room)
= 44 kilograms
The 588-gram number says that it’d get foggy in here if the moisture content ever got much above (588 g)/(44000 g) = 1.3% by mass, which sounds reasonable.
88 milliliters ain’t much, so how come the unit used up a liter of water in just one day?
Ah-hah. Air’s not sitting still. Ventilation system continually brings in low‑humidity outside air. Plus, clients complain in the wintertime about cold drafts leaking in around the door, transom and windows. If it weren’t for air shuttling in and out, we’d use up all the oxygen in here — that isn’t happening. Wonder how to calculate that flow. Bottom line is, humidifier doesn’t moisten what’s in the room so much as it loads up what comes in dry.
Problem — evaporating water cools air, Old Reliable says 2256 kilojoules per kilogram. 88 grams won’t have much effect, but a liter/day is a kilogram/day. An hour is 1/24 of a day. 1/24 liter means 94 kilojoules per hour of cooling. Air heat capacity is 1 kilojoule/(kilogram oC). (94 kJ/44 kg air)=2.1 degrees per hour. Suddenly I feel chilly.
~~ Rich Olcott