“Mom! Uncle Sy! Come outside NOW before it goes away!”
“Whah— oooh!”
”An aurora! Thanks for calling us.”
“Glowing curtains rippling across the sky! Spotlights shining down through them! Where do those come from?”
“From the Sun, Teena.”
“C’mon, Sy. The Sun’s 93 million miles away. Even if that bright streak up there is as much as 10 miles across, which I doubt, the beam from the Sun would be only a teeny‑tiny fraction of a degree wide. Not even magnetars send out anything that narrow.”
“Didn’t say it’s a beam, Sis. The whole display comes from the Sun as single package. Sort of. Sometimes.”
“Even for you, little brother, that’s a new level of weasel‑wording.”
“Well, it’s complicated.”
“So unravel it. Start from the beginning.”
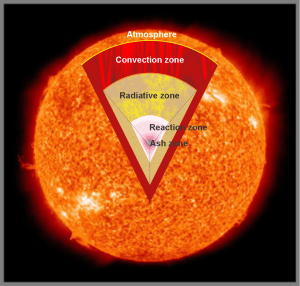
“Okay. The Sun’s covered in plasma—”
“Eww!”
“Not that kind of plasma, Teena. This is mostly hydrogen atoms except they’re so hot that the electrons and protons break away from each other and travel separately. What have they told you in school about magnets?”
“Not much. Umm … electric currents push on magnets and that’s how motors work, and magnets push on electrons and that’s how a generator works. Oh, and Mr Cox laid a sheet of paper on top of a magnet and sprinkled iron filings on it so we could see the lines of force, but when I asked him what made the magnetism ’cause I didn’t see any wires he started talking about electrons in iron atoms and then the bell rang and I had to go to Spanish class.”

<sigh> “The clock rules, doesn’t it? Anyway, he was on the right track, but I want to get back to those lines of force. Were they there before he sprinkled on those filings?”
“Mmm … Mom would say, ‘That’s a good question,’ but how could you know? I’m gonna say they were.”
“Your Mom would be right, but sorry, you’re wrong. With no iron filings in the picture, the magnetic field is nice and smooth, everywhere just the same or maybe only a little bit stronger or weaker than neighboring points. No lines. Conditions change when you put the first bit of iron anywhere in the field. As Mr Cox was probably saying when the bell interrupted, the electrons in the grain’s iron atoms align orbitals with the magnetic field. The alignment affects the surrounding field and that pulls in other iron bits that change the field even more.”
“But wouldn’t that make just a solid iron blob?”
“No, because a magnetic field has both strength and direction. Once the first particle points along the field, the iron bits it recruits rotate to point mostly in the same direction. You wind up with a chain of specks tracing out where they’ve acted together to alter the field. The chain’s surrounded by spaces where the field’s been stressed.”
“And then lotsa chains make lotsa lines, yeah!”
“I see where you’re headed, Sy. You’re going to claim that the vertical lines we see in the curtains trace out the Sun’s magnetic field.”
“Not quite, Sis. There’s only one magnetic field, a combination of Earth’s field, the Sun’s field, and the magnetic fields contained in whatever the Sun throws our way. Way out here Earth’s field is about ten thousand times stronger than the Sun’s is, but the fields inside a CME can range up to 10% or 20% of Earth’s. The moving curtains up there are the result of a magnetic tussle between us and a CME or maybe a flare’s outflow.”

Credit: NASA/GSFC/SOHO/ESA
“But there aren’t any iron filings up there, Uncle Sy!”
“True, but there are free charged particles in the ionosphere thanks to UV radiation from the Sun. A free electron caught in a magnetic field whips into a tight spiral. Its field gets neighbor particles spiraling. Pretty soon you wind up with a chain of them spiraling together, lining up like the filings do.”
“The spotlights?”
“Probably ion blobs embedded in the CME, but that’s a guess.”

licensed under CC BY-SA 4.0
~ Rich Olcott