Between COVID and the post‑holiday wind‑down, things are slow. Vinnie and I are playing cards on my office side table, except my only deck is missing the heart face cards (long story) so we’re just trying to edge‑stack them. It’s not going well. “Geez, Sy, these towers collapse so quick, it’s boring. What else you got around here?”
“Well, before you arrived I was chasing prime numbers on Old Reliable for a New Year piece. Did you know, for instance, there we’re smack in the middle of a decade-long prime year dearth?”
“Prime year dearth?”
“Prime as in not divisible by any number other than itself and one, dearth as in no year’s name being a prime number since 2017 and the next one isn’t until 2027. In the forty‑four years leading up to 2017 we averaged one prime per 5½ years. On the other hand, after 2029 (also a prime year, by the way) there’s fifty‑two years with only five primes.””
“Is there some rule for how many to expect?”
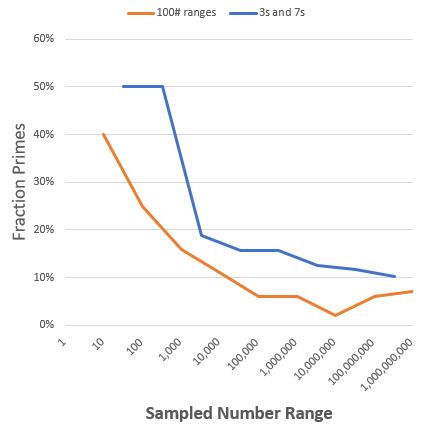
“Sort of. I sampled a series of hundred‑number ranges on up to a billion. The percentage of primes fell off as the numbers got larger, settled in at about 6%.”
“Makes sense — you got a bigger number, you got more little numbers that might divide into it.”
“Mm-hm. Something weird happens around ten million, though. The percentage drops down to only 2% but then it goes right back up to around 6% and stays there. I tried different scan resolutions but couldn’t locate any single especially long non‑prime string. The mathematicians have carried the research a lot further than my little experiment. The Prime Number Theorem gives a general curve that’s good ‘for sufficiently large numbers,’ but a million is a small number on their scale. As a physicist I’m a bit frustrated because the Theorem says, ‘This is the way it is‘ but it doesn’t give a reason. Although there probably isn’t a reason, any more than there’s a reason for 2017 being a prime to begin with.”
“I know what you mean. My car’s Owner Manual is the same way. Uhh… as I recall, you had a post a while ago about primes and 3’s and 7’s.”
“That was for New Year 2016, to be exact. Yeah, I found a collection of primes like 3337 and 733333 that have a string of 3’s or 7’s fronted and trailed by 3’s or 7’s. It wasn’t a bad bet. No primes (except 2 and 5) can have 0, 2, 4, 5, 6 or 8 as a trailing digit, right?”
“Lemme think for a minute. … Right.”
“That list didn’t include scrambled combinations like 37737, so what I did this year was to use Old Reliable to construct a big list of all possible 3’s‑and‑7’s numbers between 3 and a billion.”
“That’s a lot of numbers.”
“Not so many, actually, only about 1000. I told Old Reliable not to sample numbers that have any non‑3‑or‑7 digit buried in them somewhere. That’s a lot of pass‑overs.”
“That’s a lot of checking and skipping.”
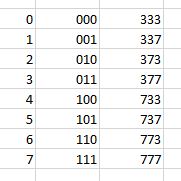
“I used a short cut. It’s easy to build a list of all possible numbers with a certain number of binary digits — just count in binary. The three‑digit binary numbers, for instance, give you every zero‑one combination between 000 is zero and 111 is seven. Then I converted all the zeroes to 3’s and all the ones to 7’s and got every 3’s‑and‑7’s number between a hundred and a thousand with no interlopers. As a bonus that method organizes the overall list by powers of ten, like 333 to 777 in a sublist, 3333 to 7777 in another and so on. I counted the primes in each sublist and charted all the sublist percentages in the same graph as the hundred‑number sampling. Pretty much the same curve, but no dip near 10 million. For the heck of it I played the same game with 1’s and 9’s. Same behavior. Oh well.”
“So that’s how you keep yourself occupied on a slow day, huh? I got a New Year prediction for you.”
“What’s that?”
“I’m gonna bring you a couple fresh decks of playing cards.”
~~ Rich Olcott