Al’s coffee shop, the usual mid-afternoon crowd of chatterers and laptop-tappers. Al’s walking his refill rounds, but I notice he’s carrying a pitcher rather than his usual coffee pot. “Hey, Al, what’s with the hardware?”
“Got iced coffee here, Sy. It’s hot out, people want to cool down. Besides, this is in honor of IceCube.”
“Didn’t realize you’re gangsta fan.”
“Nah, not the rapper, the cool experiment down in the Antarctic. It was just in the news.”
“Oh? What did they say about it?”
“It’s the biggest observatory in the world, set up to look for the tiniest particles we know of, and it uses a cubic mile of ice which I can’t think how you’d steer it.”
A new voice, or rather, a familiar one. “One doesn’t, Al.”
 “Hello, Jennie. Haven’t seen you for a while.”
“Hello, Jennie. Haven’t seen you for a while.”
“I flew home to England to see my folks. Now I’m back here for the start of the Fall term. I’ve already picked a research topic — neutrinos. They’re weird.”
“Hey, Jennie, why are they so tiny?”
“It’s the other way to, Al. They’re neutrinos because they’re so tiny. Sy would say that for a long time they were simply an accounting gimmick to preserve the conservation laws.”
“I would?”
“Indeed. People had noticed that when uranium atoms give off alpha particles to become thorium, the alpha particles always have about the same amount of energy. The researchers accounted for that by supposing that each kind of nucleus has some certain quantized amount of internal energy. When one kind downsizes to another, the alpha particle carries off the difference.”
“That worked well, did it?”
“Oh, yes, there are whole tables of nuclear binding energy for alpha radiation. But when a carbon-14 atom emits a beta particle to become nitrogen-14, the particle can have pretty much any amount of energy up to a maximum. It’s as though the nuclear quantum levels don’t exist for beta decay. Physicists called it the continuous beta-spectrum problem and people brought out all sorts of bizarre theories to try to explain it. Finally Pauli suggested maybe something we can’t see carries off energy and leaves less for the beta. Something with no charge and undetectable mass and the opposite spin from what the beta has.”
“Yeah, that’d be an accounting gimmick, alright. The mass disappears into the rounding error.”
“It might have done, but twenty years later they found a real particle. Oh, I should mention that after Pauli made the suggestion Fermi came up with a serious theory to support it. Being Italian, he gave the particle its neutrino name because it was neutral and small.”
“But how small?”
“We don’t really know, Al. We know the neutrino’s mass has to be greater than zero because it doesn’t travel quite as fast as light does. On the topside, though, it has to be lighter than than a hydrogen atom by at least a factor of a milliard.”
“Milliard?”
“Oh, sorry, I’m stateside, aren’t I? I should have said a billion. Ten-to-the-ninth, anyway.”
“That’s small. I guess that’s why they can sneak past all the matter in Earth like the TV program said and never even notice.”
This gives me an idea. I unholster Old Reliable and start to work.
“Be right with you… <pause> … Jennie, I noticed that you were being careful to say that neutrinos are light, rather than small. Good careful, ’cause ‘size’ can get tricky at this scale. In the early 1920s de Broglie wrote that every particle is associated with a wave whose wavelength depends on the particle’s momentum. I used his formula, together with Jennie’s upper bound for the neutrino’s mass, to calculate a few wavelength lower bounds. Momentum is velocity times mass. These guys fly so close to lightspeed that for a long time scientists thought that neutrinos are massless like photons. They’re not, so I used several different v/c ratios to see what the relativistic correction does. Slow neutrinos are huge, by atom standards. Even the fastest ones are hundreds of times wider than a nucleus.”
Momentum is velocity times mass. These guys fly so close to lightspeed that for a long time scientists thought that neutrinos are massless like photons. They’re not, so I used several different v/c ratios to see what the relativistic correction does. Slow neutrinos are huge, by atom standards. Even the fastest ones are hundreds of times wider than a nucleus.”
“With its neutrino-ness spread so thin, no wonder it’s so sneaky.”
“That may be part of it, Al.”
“But how do you steer IceCube?”
~~ Rich Olcott





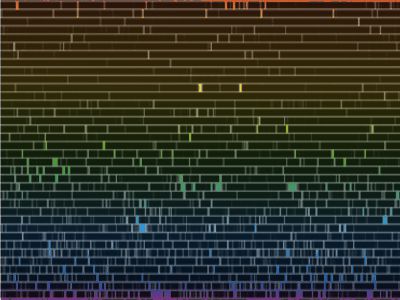 “You’re right, Sy. It’s not a particularly pretty picture, but it shows that nice strong sodium doublet in the yellow and the broad iron and hydrogen lines down in the green and blue. I’ll admit it, Vinnie, this is a faked image I made to show my students what the solar atmosphere would look like if you could turn off the photosphere’s continuous blast of light. The point is that the atoms emit exactly the same sets of colors that they absorb.”
“You’re right, Sy. It’s not a particularly pretty picture, but it shows that nice strong sodium doublet in the yellow and the broad iron and hydrogen lines down in the green and blue. I’ll admit it, Vinnie, this is a faked image I made to show my students what the solar atmosphere would look like if you could turn off the photosphere’s continuous blast of light. The point is that the atoms emit exactly the same sets of colors that they absorb.”





 “Hey, that’s the
“Hey, that’s the 








 Jennie’s turn — “Didn’t the chemists define away a whole lot of entropy when they said that pure elements have zero entropy at absolute zero temperature?”
Jennie’s turn — “Didn’t the chemists define away a whole lot of entropy when they said that pure elements have zero entropy at absolute zero temperature?”

